CHIMERIC HUMAN PROTEINS AND THEIR USE IN IDENTIFYING NOVEL ANTI-DEUBIQUITINASE COMPOUNDS
This technology involves chimeric proteins having deubiquitinase activity and methods of identifying anti-deubiquitinase compounds using the chimeric proteins. These methods and assays can be adapted to high throughput screening procedures to assay for anti-cancer drugs effecting deubiquitinase activity. Disrupting the Rpn11 deubiquitinase function is a validated strategy for treatment of human cancers.
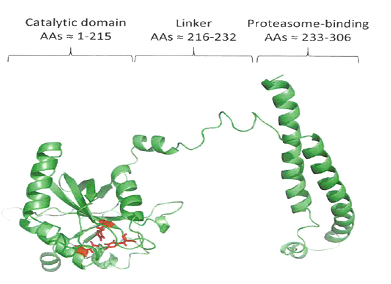
The assay utilizes a collection of genetically modified yeast strains producing a chimeric human proteasomal Rpn11 deubiquitinase. In baker’s yeast, deletion of the endogenous yeast Rpn11 gene is lethal, but inhibition of Rpn11 deubiquitinating activity is not. Inhibition of Rpn11 activity is lethal only when a second deubiquitinase, UBP6 (USP14 in humans), is deleted. Importantly, this synthetic lethal relationship is maintained in yeast harboring hRpn11. Thus, a selective inhibitor of human Rpn11 would be lethal in the hRpn11 strain lacking UBP6, but nontoxic in a strain containing UBP6 and harboring inactivating point mutations in hRpn11.By measuring cell growth in the presence of potential inhibitors using any commercially available plate reader, a high throughput cell-based screen for compounds selectively inhibiting hRpn11 can be enacted.