Facile Conversion of Red Phosphorous to Soluble Polyphosphide Anions by Reaction with Potassium Ethoxide
- Pending
Activation of phosphorus is an important process for the preparation of semiconductors and low-dimensional electronic materials. The industry, in general, uses white phosphorus, which is hazardous and should be stored under water due to its spontaneous flammability in air. Activation of red phosphorus, which is a more stable polymorph of the element, is usually done by high-temperature reactions with metals in sealed evacuated tubes. However, this process is expensive and difficult to scale up.
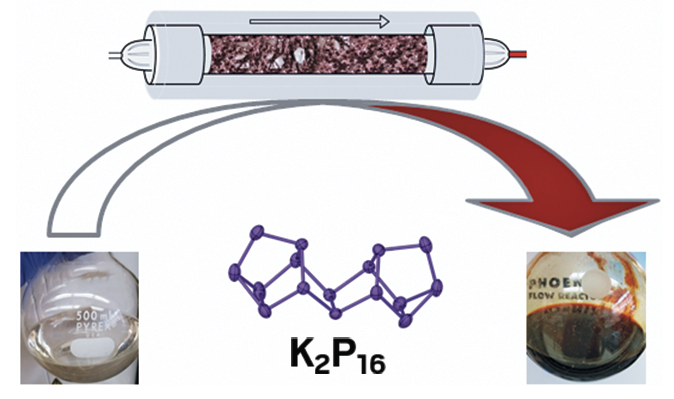
We have discovered a methodology to activated red phosphorus using inexpensive potassium ethoxide in ethanol. The reaction can be performed with mild heating and provides access to soluble polyphosphide species, which can be used to explore further chemistry of phosphorus in solution, without the need to use white phosphorus. Moreover, we showed that this process can be easily scaled up using flow chemistry approaches.
For more information: Chemists Discover a Safe, Green Method to Process Red Phosphorus
red-wonder FSU chemists pave the way of phosphorus revolution
Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201511186