Microfluidic Sample Preparation Device for Electron Microscopy
Cryogenic electron microscopy (cryoEM) is quickly becoming a routine method in the determination of high-resolution structures of biological molecules. However, for most samples before cryoEM data can be collected, the sample quality and heterogeneity must first be characterized using negative staining. Conventionally, EM grids are prepared by hand and, as such, variability is introduced due to user-to-user differences. The variability of the staining can have large effects on the final stained sample, ultimately hindering the resolution, image processing, and data analysis.
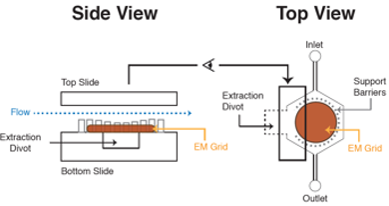
A microfluidic platform is presented for preparing negatively stained grids for use in transmission electron microscopy (EM). The microfluidic device is composed of glass etched with readily fabricated features that facilitate the extraction of the grid post staining and maintains the integrity of the sample. The device allows for sealing of an electron microscopy grid, facile and reproducible delivery of a sample, followed by delivery of subsequent solutions that could be negative stains or other biological samples. The device houses the EM grid in an outlined chamber with an access point below the grid for gentle and easy recovery of the EM grid. The fluid is directed to the grid using the integrated channels of the microfluidic system.
Utilization of this device simultaneously reduced environmental contamination on the grids and improved the homogeneity of the heavy metal stain needed to enhance visualization of biological specimens as compared to conventionally prepared EM grids.
High-magnification images from grids prepared by the microfluidic system showed similar image qualities as those prepared by hand. With this methodology for housing the grid, opportunities are abound for more integrated systems using elastomeric materials for incorporation of valving and other microfluidic features. For example, this system can subsequently be complemented with gradient generators or multianalyte perfusion and reaction timers to study both multivariable interactions as well as reaction kinetics. This proof of principle paves the way for future added layers of complexity that can be used to uniquely investigate structural biology dynamics.
Results have been published in Analytical Chemistry (Roper, 2016, American Chemical Society Publications) and led to multiple requests by research groups offering to beta test the prototype.
Advantages:
- User friendly
- Reproducibility
- Parallel/high throughput
- Straightforward manufacturing
For further reading, please visit: