Stereo Controlled Synthesis of (E,Z)-Dienals via Tandem Rh(i) Catalyzed Propargyl Claisen Rearrangement
Share:
Tech ID:
14-160
Principal Investigator:
Dr. Igor Alabugin
Licensing Manager:
Description:
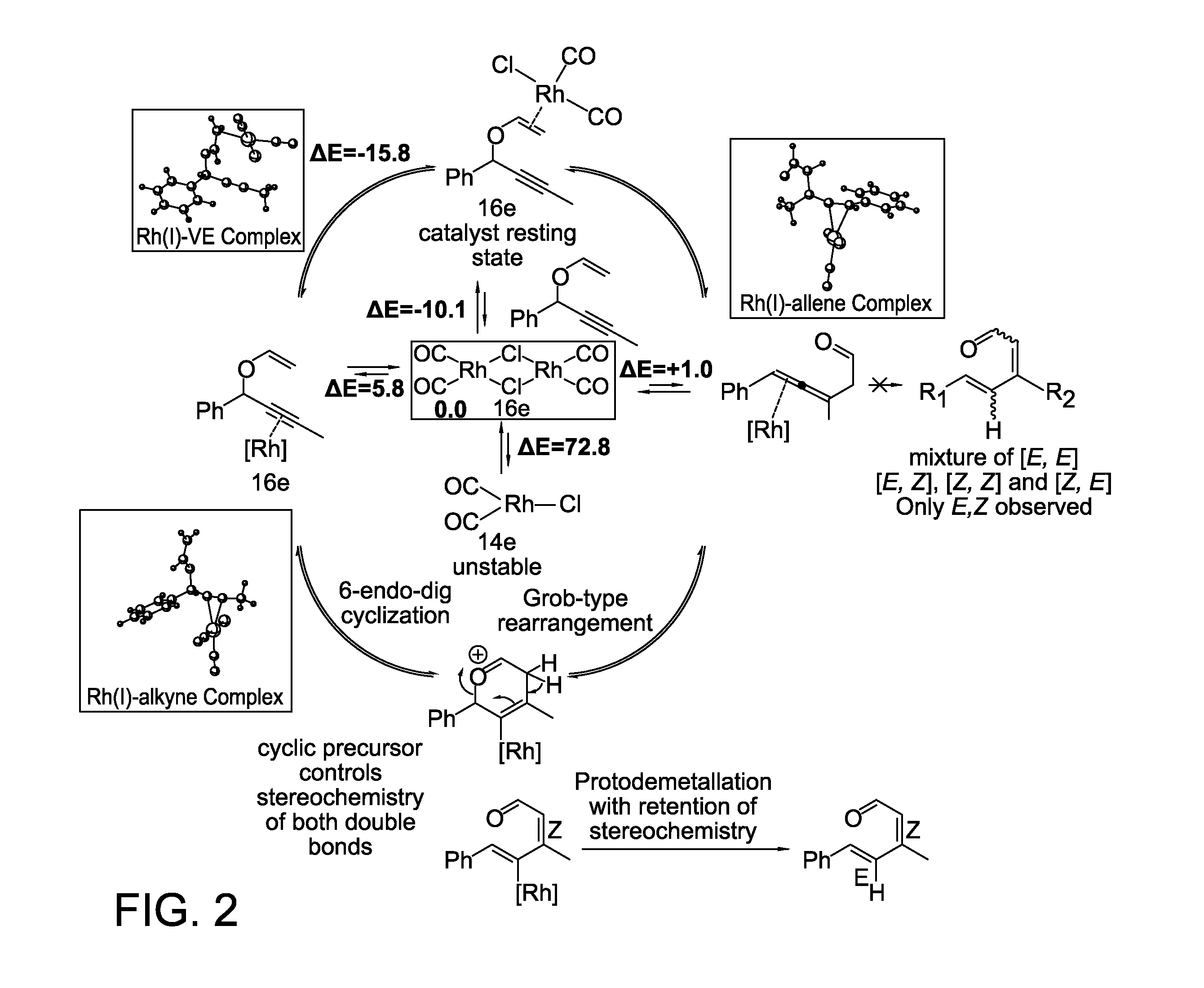
A novel Rh(I)-catalyzed approach to synthesizing functionalized (E,Z) dienal compounds has been developed via tandem transformation where a stereoselective hydrogen transfer follows a propargyl Claisen rearrangement. Z-Stereochemistry of the first double bond suggests the involvement of a six-membered cyclic intermediate whereas the E-stereochemistry of the second double bond stems from the subsequent protodemetallation step giving an (E,Z)-dienal.