Stimulus Triggered Recyclable Catalysts
N-heterocyclic carbene ("NHC") compounds are often used as supporting ligands in homogeneous organometallic catalysts. NHC-based catalysts can catalyze a wide range of reactions, including metathesis reaction, Suzuki reactions, Negishi coupling reactions, Buchwald-Hartwig amination reactions, and organo-catalytic reactions among many others.
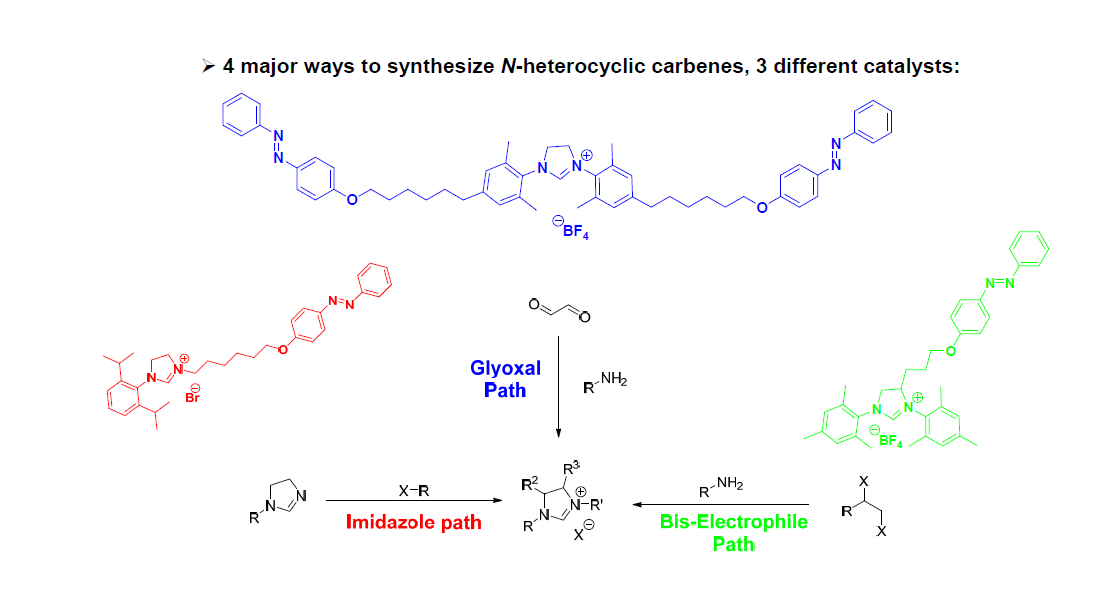
This invention shows that host-guest interactions can be used to recover/recycle well-defined organometallic catalysts. The host-guest interaction is a strong molecular recognition (non-covalent bonding) based on supramolecular chemistry. The N-heterocyclic carbene (NHC) is an excellent supporting ligand for homogeneous organometallic catalysis. Various azobenzene-tagged NHCs are proposed to test the resulting catalyst's activity and recover/recycle capability. The basic principle is that a homogeneous catalyst can be prepared with NHC ligand that has azobenzene group. After a designated chemical reaction, the catalyst can be recovered by 13-cyclodextrin (CD) via host-guest reaction . Note that the catalyst having azobenzene group doesn't engage to CD, if there is no stimulus. Thus, the catalyst can be selectively and conveniently recovered by external stimulus. Those external stimulus includes light and electricity. Also the azobenzene-tagged NHCs can be used in organo-catalytic reactions.
Previously, there were two main approaches to recycle/recover the catalyst. The first approach is immobilization of a heterogeneous tag on a catalyst structure. This method allows for easy separation (filtration), but it suffers from severely low catalytic activity. The second strategy is having a switchable tag on a homogeneous catalyst to recover the catalyst in a heterogeneous state (solid) after a desired chemical reaction. This method requires costly processes to trigger phase changes.
Advantages
- Catalyst is homogeneous
- Phase changes are not necessary
- Easy recovery process
- Repeatedly reusable
- Cost efficient